- Shapiro J. Clinical practice. Hair loss in women. N Engl J Med. 2007;357:1620-30.
- Norwood OT. Incidence of female androgenetic alopecia (female pattern alopecia). Dermatol Surg. 2001;27:53-4.
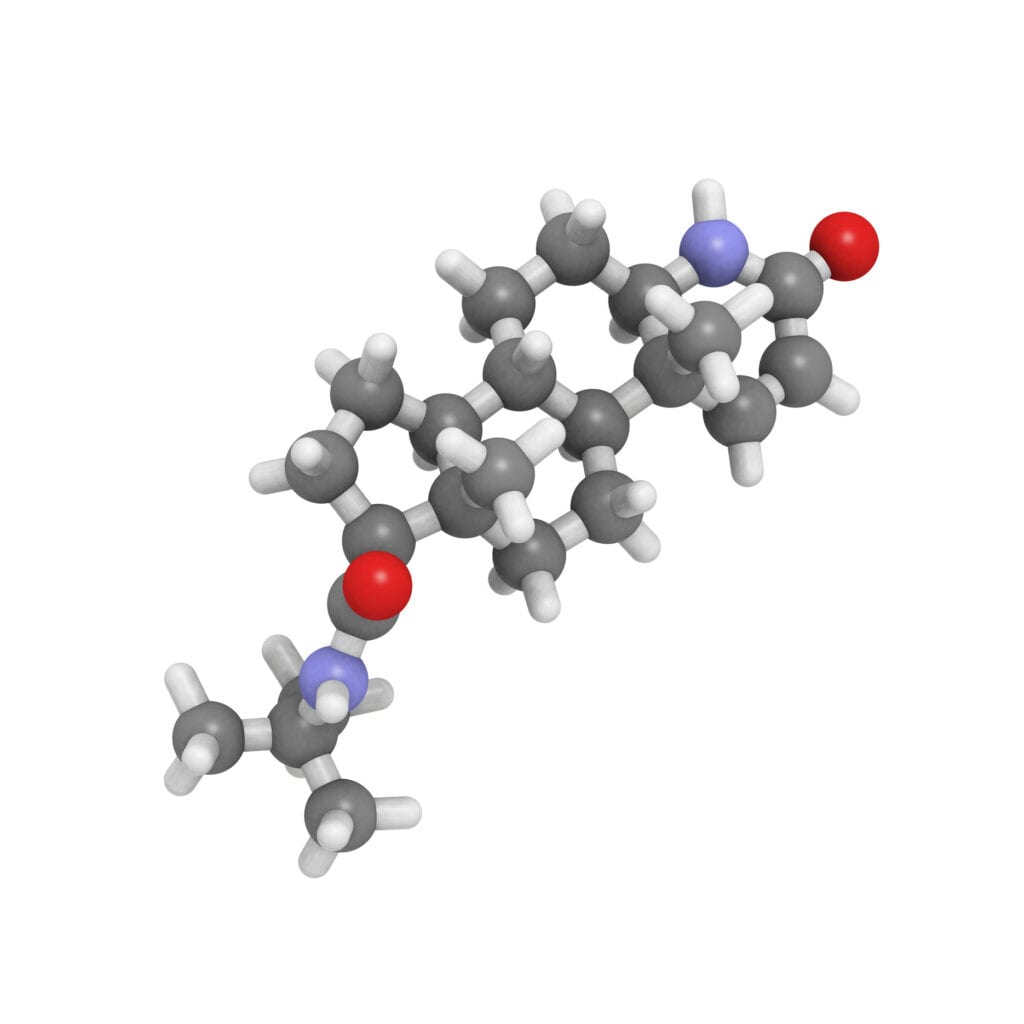
- Messenger AG, Rundegren J. Minoxidil: mechanisms of action on hair growth. Br J Dermatol. 2004 Feb;150(2):186-94. Review. PubMed PMID: 14996087.
- Johnson & Johnson Consumer and Personal Products Worldwide. A phase 3 multi-center parallel design clinical trial to compare the efficacy and safety of 5% minoxidil foam vs. vehicle in females for the treatment of female pattern hair loss (androgenetic alopecia). In: ClinicalTrials.gov, Identifier: NCT01226459. Last updated June 3, 2014. Available at: http://clinicaltrials.gov/ct2/show/results/NCT01226459?term=minoxidil§=X4301256#othr. Accessed September 1, 2014.
- Johnson & Johnson Consumer and Personal Products Worldwide. A phase 3 multi-center parallel design clinical trial to compare the efficacy and safety of 5% minoxidil foam vs. 2% minoxidil solution in females for the treatment of female pattern hair loss – androgenetic alopecia. In: ClinicalTrials.gov, Identified: NCT01145625. Last updated May 19, 2014. Available at: http://clinicaltrials.gov/ct2/show/study/NCT01145625?term=minoxidil§=X430126. Accessed September 1, 2014.
- Blume-Peytavi U, Hillmann K, Dietz E, et al. A randomized, single-blind trial of 5% minoxidil foam once daily versus 2% minoxidil solution twice daily in the treatment of androgenetic alopecia in women. J Am Acad Dermatol. 2011 Dec;65(6):1126-34 e2.
- Van Zuuren EJ, Fedorowicz Z, Schoones J. Interventions for female pattern hair loss. Cochrane Database Syst Rev. 2016 May 26 (5):CD007628. doi:10.1002/14651858.CD007628.pub4. Review. PubMed PMID: 27225981; PubMed CentralPMCID: PMC6457957.
- Rathnayake D, Sinclair R. Innovative use of spironolactone as an antiandrogen in the treatment of female pattern hair loss. Dermatol Clin. 2010Jul;28(3):611-8. doi: 10.1016/j.det.2010.03.011. PubMed PMID: 20510769.
- Sinclair R, Wewerinke M, Jolley D. Treatment of female pattern hair loss with oral antiandrogens. Br J Dermatol. 2005 Mar;152(3):466-73. PubMed PMID: 15787815.
- Stout SM, Stumpf JL. Finasteride treatment of hair loss in women. Ann Pharmacother. 2010 Jun;44(6):1090-7. doi: 10.1345/aph.1M591. Epub 2010 May 4. Review. PubMed PMID: 20442354.
- Paradisi R, Porcu E, Fabbri R, Seracchioli R, Battaglia C, Venturoli S. Prospective cohort study on the effects and tolerability of flutamide in patients with female pattern hair loss. Ann Pharmacother. 2011 Apr;45(4):469-75. doi: 10.1345/aph.1P600. Epub 2011 Apr 12. PubMed PMID: 21487083.
- Yazdabadi A, Sinclair R. Treatment of female pattern hair loss with the androgen receptor antagonist flutamide. Australas J Dermatol. 2011 May;52(2):132-4. doi: 10.1111/j.14400960.2010.00735.x. Epub 2011 Mar 1. PubMed PMID: 21605098.
- Jimenez JJ, Wikramanayake TC, Bergfeld W, Hordinsky M, Hickman JG, Hamblin MR, Schachner LA. Efficacy and safety of a low-level laser device in the treatment of male and female pattern hair loss: a multicenter, randomized, sham device-controlled, double-blind study. Am J Clin Dermatol. 2014 Apr;15(2):115-27. doi: 10.1007/s40257-013-0060-6. PubMed PMID: 24474647; PubMed Central PMCID:PMC3986893.
Moran LJ, Hutchison SK, Norman RJ, Teede HJ. Lifestyle changes in women with polycystic ovary syndrome. Cochrane Database Syst Rev. 2011 Feb 16;(2):CD007506. doi: 10.1002/14651858.CD007506.pub2. Review. Update in: Cochrane Database Syst Rev. 2011;(7):CD007506. PubMed PMID: 21328294.
Lake Oswego, Oregon 97034